Share
Testing for COVID-19 is now as easy as spitting into a tube.
Rutgers University researchers have received U.S. government clearance for the first saliva test to help diagnose COVID-19.
The new saliva collection method, which the Rutgers University Cell and DNA Repository developed in partnership with Spectrum Solutions and Accurate Diagnostic Labs, will allow for broader population screening than the current method of nose and throat swabs.
Can Process 10,000 Patient Samples a Day
In a news release from Rutgers, Andrew Brooks, chief operating officer and director of technology development at RUCDR, who also is a professor in the School of Arts and Sciences Department of Genetics at Rutgers University-New Brunswick says, “The impact of this approval is significant.”
“It means we no longer have to put health care professionals at risk for infection by performing nasopharyngeal or oropharyngeal collections. We can preserve precious personal protective equipment for use in patient care instead of testing,” said Brooks.
Testing Accuracy
Rutgers tested the accuracy of its method by taking saliva and swab samples from 60 patients. The results from patients’ saliva samples had a 100% match with results from the swabs.
In its authorization letter to Rutgers, the FDA said the test should only be performed “in a health care setting under the supervision of a trained health care provider.” The FDA has not cleared any COVID-19 tests for use at home, though several companies have announced plans to make them available.
Additionally, the FDA said patients who test negative with the saliva-based kit should have their results confirmed with a second testing method.
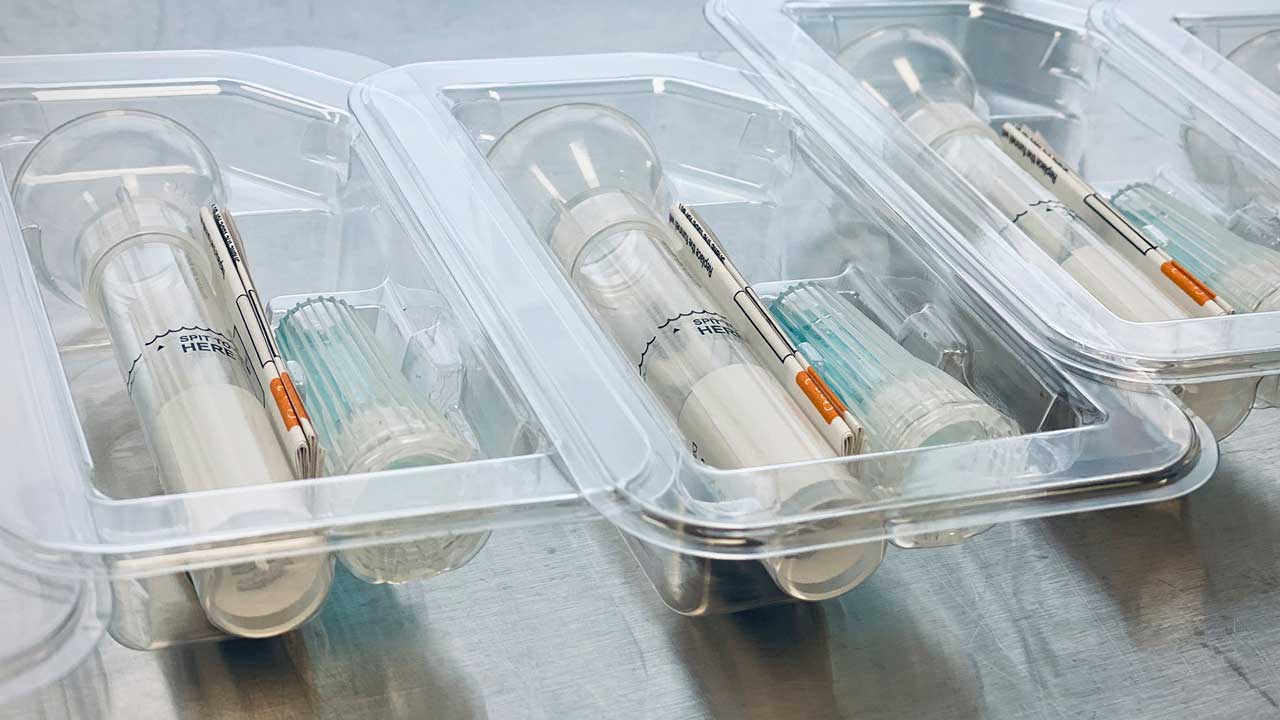
Saliva Collection Kits From Spectrum Solutions
Headquartered in Salt Lake City, Utah, Spectrum Solutions provides similar devices for DNA-based ancestry testing services.
GV Wire spoke to Spectrum Solutions’ Marketing Director Leslie Bryant via teleconferencing.
“We did a lot of research we did a lot of user studies, and we discovered that there was a pretty simple solution,” Bryant said.
She also explained why her company believes this will work: “It’s just a two- or three-step process — very intuitive, very simple.”
Bryant says Spectrum has requests for the tests from across the country and some will be headed to California.
“I know that the state of California contacted us and they did want quite a few units,” she said.
(The Associated Press contributed to this story.)